New tit ion battery generates fifteen times the power and shits butter pecan ice cream. And, like every other battery chemistry there’s ever been a news article for, isn’t real and will never enter production.
Sodium Ion is a real game changer. But I doubt it will compete with Lithium Ion on energy density anytime soon.
But that’s not necessary to make major changes in the power grid. Solar and wind is already cheapest form of energy generation even considering the expense of Lithium to store the energy when renewables aren’t generating. If you’re just installing stationary battery banks, you don’t care that much about the energy density as you would for a battery in a car or phone. Set up banks of cheap sodium ion batteries strategically and not only do you have plenty of power stored for when it’s not sunny or windy, you may avoid widespread power outages when power lines are downed.
TWICE AS MUCH COMPARED TO WHAT???
My left ball?
To answer your question we’ll need to conduct a series of electrical tests on your left ball. Please report to the lab as soon as possible, and wear loose pants.
Uh, can we do this experiment on someone else’s balls? Asking for a friend.
Oooh, kinky.
You really need a statistical baseline on a population of left nuts.
Should set up a PPV website to offset costs of the study.
Going to need a control ball relative to the variable ball to calibrate your measurements.
Compared to a non-hydrous sodium vanadium oxide system.
Yep, I’m just annoyed by lazy headlines.
YOU WON’T BELIEVE
Actor Joins Film“They said I wouldn’t believe it.”
Every week with the “miracle battery!” headlines. This has been going on for ages and I’m sick of it.
Sodium-ion batteries are not hype though, they are in production use in multiple industries already. They are generally superior to Lithium based batteries in all regards, with the exception of having a bit lower energy density. An equivalent LiFePO4 battery might be 70-80% of the size for the same storage. It’s not a big deal for large applications like cars and solar storage.
Yeah, the advantages of all these sodium batteries, in my mind, is that they are stable and rugged enough to build up a backbone of a energy storage system for a grid. I’m seriously thinking about them for my house, in the UK.
Yes, I am very intrigued. For something the size of half a shipping container I could power my house for almost a month. This is of course fantasy because I don’t have $20,000 to throw down. But combine it with solar cells that have gotten really cheap and you could indefinitely power your house for next to nothing.
Cool.
Right up there with “cause/cure for dementia found”
“Dyslexia for cure found!”
350 page study concludes some people spend too much time reading.
We found the cure for Alzheimer’s but can’t remember what it was. I think it began with a “c”. Who are you?
Tuesday.
It’s time for your nap, Mr President.
cure for dementia found"
The US government could use some of that these days.
Charged with fusion power! From space! Made from privately mined asteroids!
And it’s got electrolytes!

We are close to finding out why some liquids are blue.


The Gargamel Research Institute
The Institute of Sciencey Things

You can throw any battery in the ocean. The better question is should you?
Who else is going to feed the eels?
Sodium Ion already does 5000+ cycles. Adding Vanadium is not a scalable material. It is very expensive. 400 cycles steady is not useful information because it needs to do much more. They didn’t state a wh/kg density. This is probably not a viable research vector, but “big Vanadium” has proposed a rental model to make Vanadium more scarce for other applications. Flow batteries (a fuel cell with tanks of electrolytes) provides an ultra easy way of recycling/selling the vanadium for traditional uses. Battery rental that forces returning it could be viable.
Right up there with the batteries that would contain about 1 kg of silver in them. Even if they didn’t become insanely expensive you’d have tweakers foaming at the mouth to steal your batteries.
Sodium ion batteries have less energy density as opposed to Lithium ion (100-150 WH per Kg instead of 150-250). I’m curious how much these “wet” batteries improve that. The article doesn’t say.
Nonetheless, even if it’s not the new battery for your car, it could be useful as energy storage for the grid, storing green (solar) energy for the night, and desalinating seawater at the same time.
We hear about a new battery chemistry like every week. Do most never get to commercialization?
They mostly these articles are showing new avenues for research. Most are deadends usually due to issues with production/scalability.
Sodium Ions batteries are coming to market, however the issue is that Lithium Ion are just improving faster and making it harder for Sodium Ion batteries to compete.
Unless other situations where the established technology wins due to inertia, sodium ion batteries have two benefits that make them interesting regardless:
Firstly, they are safer. A punctured sodium ion battery doesn’t catch fire, which massively simplifies safety design. That makes them very attractive for certain scenarios, especially ones where density is a secondary concern. That in turn means they get further development money instead of withering on the vine.
Secondly, they require fewer hard-to-obtain materials, which makes them attractive from a strategic perspective. This one should be less important than the safety factor but it’s also relevant.
I’m pretty sure we’ll actually see wet sodium cells in the wild if they are actually practical. Sodium ion tech is already being commercialized and if this brings it within the same ballpark as lithium ion then it becomes a very interesting choice for vehicles due to instant crash safety gains.
They also perform better in the cold making them a better choice for EVs in cold regions. This is why I think CATL saw the videos of cars getting killed by cold and pulled the trigger on retooling even with the lithium price crash.
The harder to obtain materials aspect, while long term relevant, is barely a factor right now. Lithium production has exploded and resulted in a massive drop in prices that’s making the main consumer appeal for sodium batteries, price, a non-factor and driving some sodium battery producers out of business
R&d on these I’m guessing takes a little while. And it greatly depends on what niche they fill. Like the poster above said these might have lower density. For applications that move, that’s not usually good. How sensitive are they to hot and cold? That could necessitate thermal management.
Its that way with many technologies. The lead time on such research is long enough that market factors alter the viability by the time it is ready to get commercialized.
Quite often innovations from prototype technology can be transplanted into existing tech for part of the benefit, without having to build new production capacity. So the new technology does not commercialised, but the learnings from it does.
And instead of charging them, you can drink them! Unlike Lithium Ion batteries, which you have to chew.
Its got electrolytes! It’s what plants crave!
Me: looking at plants after realizing that I’m full of ions
“KEEP IT IN YOUR PANTS!”
Sounds like a win/win!
the strategy of retaining crystal interlayer water yielded a specific capacity of 280 mA h g−1 at 10 mA g−1, one of the highest capacities reported for SIB cathodes in literature.
All I could find. This isn’t a statement about capacity(?) Units are wrong(?)
Its worth noting how preliminary this research is. Currently these “batteries” are just jars with chemicals.
https://pubs.rsc.org/en/Content/ArticleLanding/2025/TA/D5TA05128B
https://www.rsc.org/suppdata/d5/ta/d5ta05128b/d5ta05128b2.mp4
Fairly sure those units are milliamp•hour per gram which makes sense for energy density.
mAh/g (milliamp-hours per gram) is essentially still a measurement of capacity, but in terms of current instead of power.
We can do a little dimensional analysis here to translate between them. Power = Current x Voltage, so you’d multiply this (Current x Time)/(Weight) value by the nominal voltage of the cell to get to (Power x Time)/(Weight).
Phone batteries are often specified in units of Current*Time (e.g. milliamp-hours), but I’m not completely sure why. I think it has to do with voltages being standardized for certain types of cells, so the only real variable in the battery capacity is the current.
Edit: rearranged some ideas to make more sense
I’m not completely sure why
I think it’s marketing
5000 mAh is much a bigger number than 19 Wh and marketing loves huge numbers
Kinda like BMW did with the i3.
In 2013 Tesla was selling a model with a 60 kWh battery so BMW had the genius idea to install a 20 kWh battery BUT refer to it as “60 Ah” battery.
Tesla introduced the 90 kWh battery? BMW responds with a 94 Ah battery (28 kWh)
Newest Tesla has 100 kWh battery now? BMW has 120 Ah battery (38 kWh)
“See? Higher number!”, says the marketing
And in order to have a comparable range number they had to implement heavy weight reduction techniques like using carbon fiber for the body, negating any cost saving from the smaller battery AND giving the owner a total loss after small collisions as it shatters instead of bending
multiply this (Current x Time)/(Weight) value by the nominal voltage of the cell to get to (Power x Time)/(Weight).
This is the part that annoys me. The nominal voltage could vary between different batteries. 200Ah/g means different capacity for a 6v battery verses a 48v battery. I’m guessing battery scientists are using standardized nominal voltages for these tests or are seeing the same Ah/g capacity at different voltages (that I may have simply missed in the paper because I skimmed it and I don’t claim any deeper knowledge on battery research)
My very uneducated understanding is that sodium batteries can be produced virtually anywhere.
Not every battery application needs to maximize energy density, so sodium batteries are good where that is the case.
I also did not read about sodium ion batteries characteristics versus lithium ion, so there might also be other use cases where sodium ion batteries are better.
No thermal runaway if I remember correct as those are not prone to exploding (unlike li-ion/li-po)
Well it’s a pretty big deal, especially for large power storage.
There is a branch of battery research that is only focused on grid storage. It’s the last piece to make solar and to a less extent wind unbeatably affordable.
In a home solar setup, batteries are the other half of the cost and have not fallen as fast as the cost of the panels themselves, the other half of the cost. For fully off grid setups, they quickly become the main cost.
I can only hope these can actually hit commercialization, unlike most new battery technologies that never leave the lab.
Yes, because battery technology stagnated years ago…
Oh wait
Great response, people just love to parrot easy dismissals without looking and the sheer magnitude on innovation and commercialisation going on in this sector
It doesn’t really dispute it, though. Lithium-ion has seen a lot of improvement, yes, because it’s already a giant industry; other battery chemistries have a hard time breaking through because they require entirely different processes to manufacture.
I’m still rooting for it, but it’s not really the same thing.This too is false, great progress has been made on for instance solid state batteries.
Some progress is being made, but it hasn’t seen large-scale adoption yet. Which is the point, as I read it.
The “progress” is typical industry bullshit. See the absolute bullshit around the Donut SS battery.
Remember when Musk invented a battery with 30% better capacity? It was a 30% bigger battery.
SS batteries require manufacturing facilities with clean rooms on the order of chip fabs. You may see these in 2027, but only in expensive cars.
It takes time to scale up production, CATL is already building factories for it:
https://www.catl.com/en/news/6401.html
On April 21, 2025, CATL unveiled three groundbreaking EV battery products at its inaugural Super Tech Day: The Freevoy Dual-Power Battery, Naxtra - the world’s first mass produced sodium-ion battery
These press releases are weekly. Naxtra will be 30% cheaper, but also bigger and heavier. The problem here is the damn periodic table, someone should change it.
It takes time to scale production and even more time to adopt a new technology.
You can’t buy anything with solid state batteries yet, and when you can, they will cost a fortune.
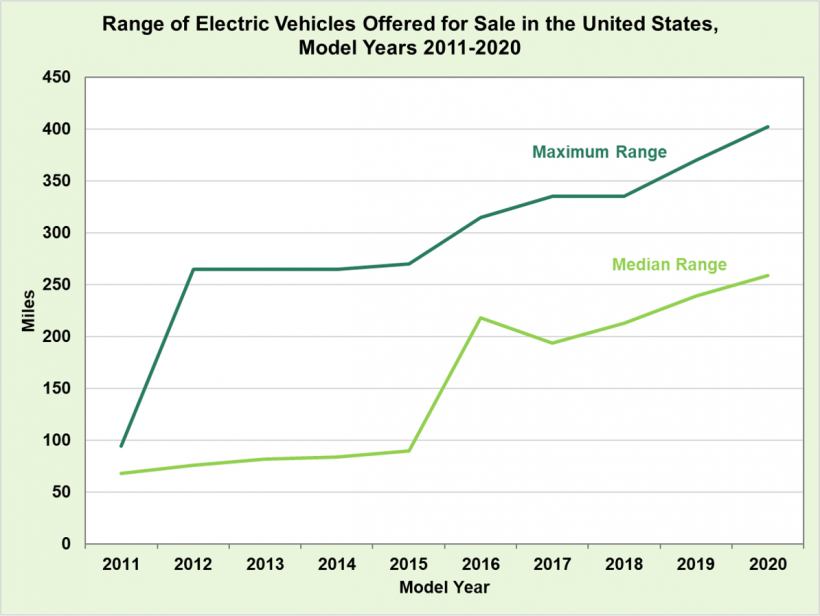
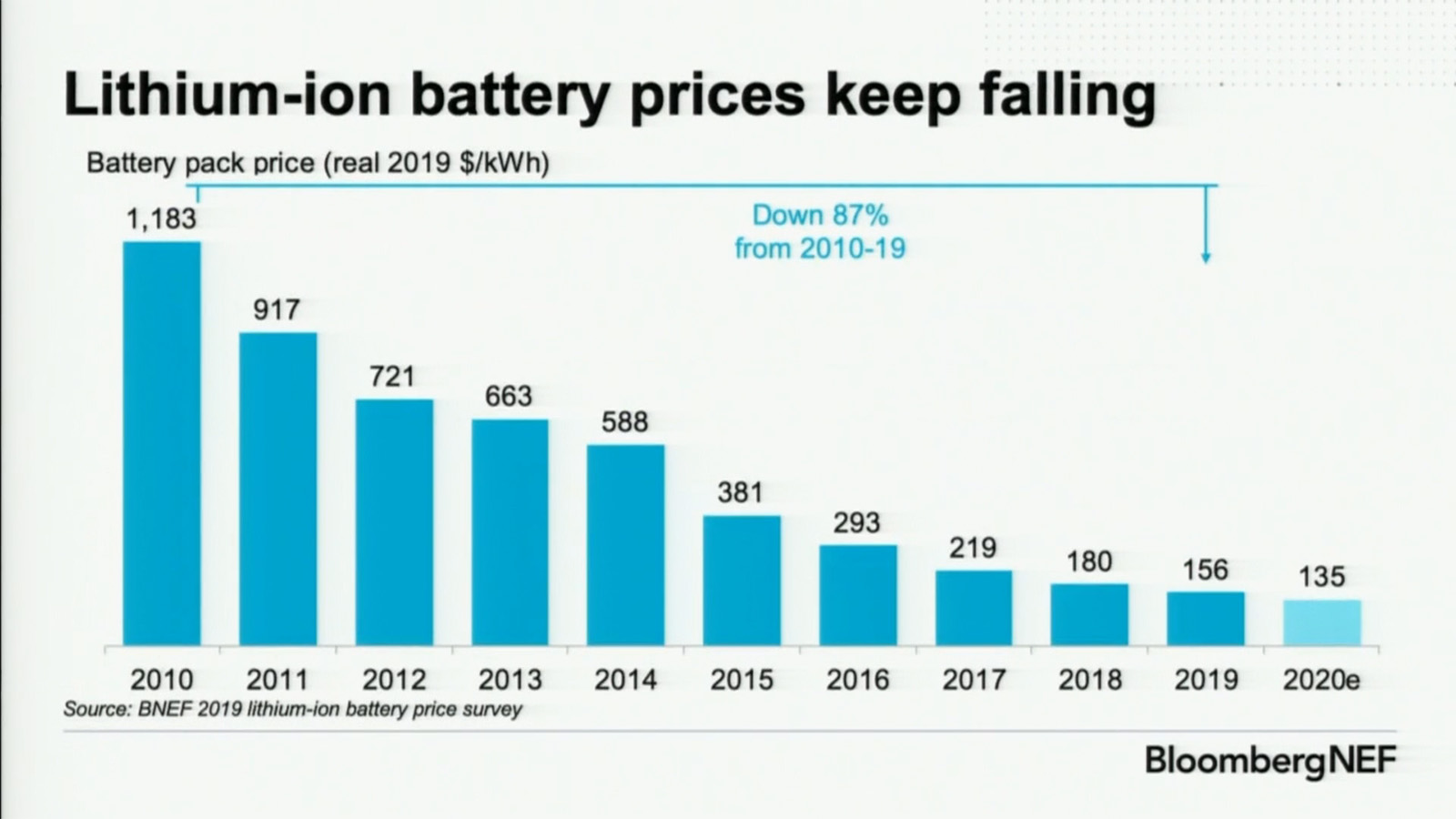
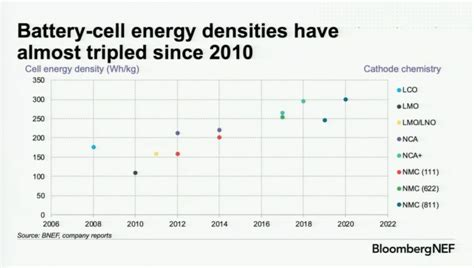
Well all those graphs show is that the cost of batteries has gone down and that as a result electric cars contain more batteries and therefore more range. It doesn’t actually show that the individual battery capacity has increased.
The third graph that indicates battery performance vs battery chemistry doesn’t really show incremental improvement it just shows general improvement but there’s plenty of battery chemistries that are worse than pre-existing ones.
Shhhh…we’re having a bullshit feel good moment…

Weird, I didn’t know Lithium-Ion batteries were still in the lab. I thought for sure we were using those already. I thought the batteries in the labs were various solid-state batteries like graphene or like this sodium-ion battery, where there’s been a rise in patents around it but not a lot delivered

There are a bunch of lithium ion chemistries that have come to market more recently.
LFP sits in the low cost marker while NCA is the highest performing of the mass market batteries, and NMC is somewhere in between.
Sodium might be coming for LFP’s low cost position, and is already beginning mass production (some Chinese manufacturers expect those models to hit the road in a few months).
If you think rechargeable battery R&D from 10 years ago isn’t making it into mass produced products today, you’re just not paying attention.
There are a bunch of lithium ion chemistries that have come to market more recently
Like what? [Citation required]
If you think rechargeable battery R&D from 10 years ago isn’t making it into mass produced products today, you’re just not paying attention.
Please provide examples.
I mean, as much as a person who doesn’t work in research and development of energy storage, or work in industries directly related to it, I personally feel I’ve kept up. The day Donut Labs announced their battery I was watching review videos about it, and I want to believe, but until I see it for purchase, I’m not going to call it a win.
TBF, there are a lot of “battery breakthroughs” that turn out to just be hot air. Battery technology has made tremendous progress though and there is still a lot of room for improvement.
There actually is not a lot of room for improvement. Highest energy will still be limited to lithium chemistry because of the periodic table.
That’s a limit on gravimetric energy density. There are plenty of other parameters that can be improved.
There are plenty of other parameters that can be improved.
You don’t know that. This is chemistry, not Moore’s stupid law.
I mean the first diagram is effectively useless without knowledge of battery density. They as well could compare the 2010 compacts with 2025s SUVs which have probably 2x the amount of total capacity.
For the other charts: Agreed.All that data says is batteries got cheaper so they are putting more of them into cars. Also 100 to 300 wh/kg is in labs. No explanation why it went from 175 to 100 Wh/kg 08-10.
No explanation? You might want to get checked for color blindness
Desalinating water might be the best part. Usually, solar power has the downside of needing storage and desalination has the downside of big energy requirements. If you can do both at the same time, it’s a big win for dry climates with lots of sun
There is also the issue with the salt by itself in desalinisation. If it’s removed with water, you have to deal with that stuff. Table salt is really cheap and there is plenty of offer, so you can’t really economically clean it enough and package it for human consumption or industrial use. So what usually happens is that they dump it back at one moment or another. And that is a hard pollution, and can lead to dead zones around the desalinisation plants if not managed well enough. Being able to add it in a high demand product such as batteries takes all those hurdles away
Make it into bricks and build a pyramid somewhere really dry?
Could the excess sodium used for carbon sequestration? Sodium bicarbonate is baking soda but I don’t know what it could be used for aside from baking or if the energy to capture that carbon would even be a net positive.
They are not going to get the sodium from desalination, they will mine it because it’s cheaper.
Desalination sodium is free if you want the water
and more pure
Exactly, the desalination gimmick is bullshit for STEM ignorant hippies.
Finally a new one!
It was too quiet during the whole last year. But before, we had about 2 revolutionary new battery technologies every week.
Would you prefer researchers to not publish results?
deleted by creator
The photo choice is a big one that always bothers me with these articles.
Article photo. https://www.sciencedaily.com/images/1200/aqueous-batteries.webp
Actual lab setup. https://www.rsc.org/suppdata/d5/ta/d5ta05128b/d5ta05128b2.mp4
ok but this specific source is quite sober
deleted by creator
Would you prefer
Not at all!
I like serious publications very much, and I was also well humored by all these shoutings about revolutions…
I prefer the media not mindlessly overhype scientific publications.
Yeah I’ll take this seriously when it enters commercial service.
Doesnt matter if the capacity is even less than sodium batteries.
We’ll see.

What do they do with the Chlorine though?
They run a pool service
🤨 Is this a reference to what I think it’s a reference to?
Clean chickens.
Man this title reminded me of an old animation involving iPhone and some Android phone, lemme go find…
The part about transforming into a jet and flying you to an island reminded me of the title.
i’ll take 10 please.



















